1
hypothetical numbers assigned to an individual atom or ion present in a substance using a set of rules
2
a substance that tends to bring about oxidation by being reduced and gaining electrons.
3
a substance that tends to bring about reduction by being oxidized and losing electrons.
5
is the slow addition of one solution of a known concentration (called a titrant) to a known volume of another solution of unknown concentration until the reaction reaches neutralization, which is often indicated by a color change
6
redox reaction that gains electrons
7
redox reaction that losses electrons
8
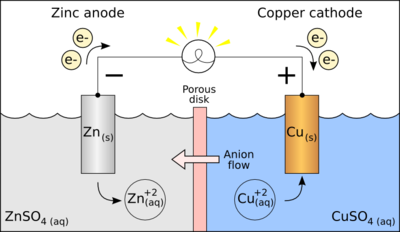
the positively charged electrode of an electrical device
9
the negatively charged electrode of an electrical device